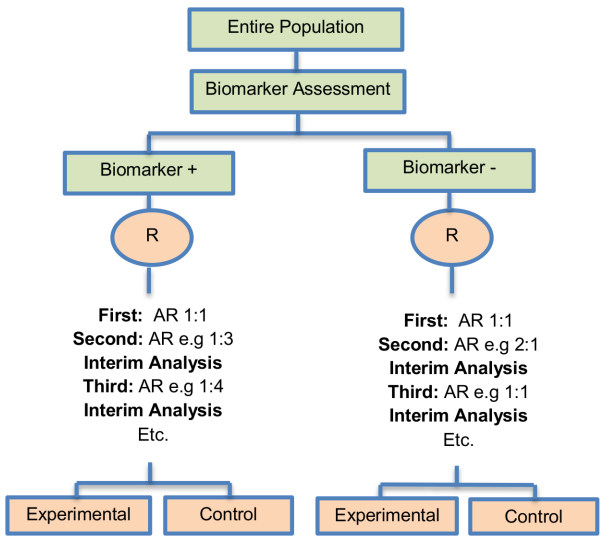
In the context of personalized medicine, this design is used when the biomarkers are only putative or not known at the beginning of a Phase II trial and is also useful when there are multiple targeted treatments and biomarkers to be considered. It aims to test simultaneously both biomarkers and treatments while providing more patients with effective therapies according to their biomarker profiles. Outcome-adaptive randomization is sometimes included under the umbrella of “Bayesian clinical trials” but as criticized by Korn and Freidlin (2011), there is nothing inherently Bayesian about it.
Alternative names: Adaptive randomization, Bayesian Adaptive, Bayesian Adaptive randomization, Combined dynamic multi-arm, Outcome-Adaptive randomization, Outcome-based Bayesian Adaptive Randomization
Adaptations: Change in randomization ratio
Variations: i) Bayesian covariate adjusted response-adaptive randomization

